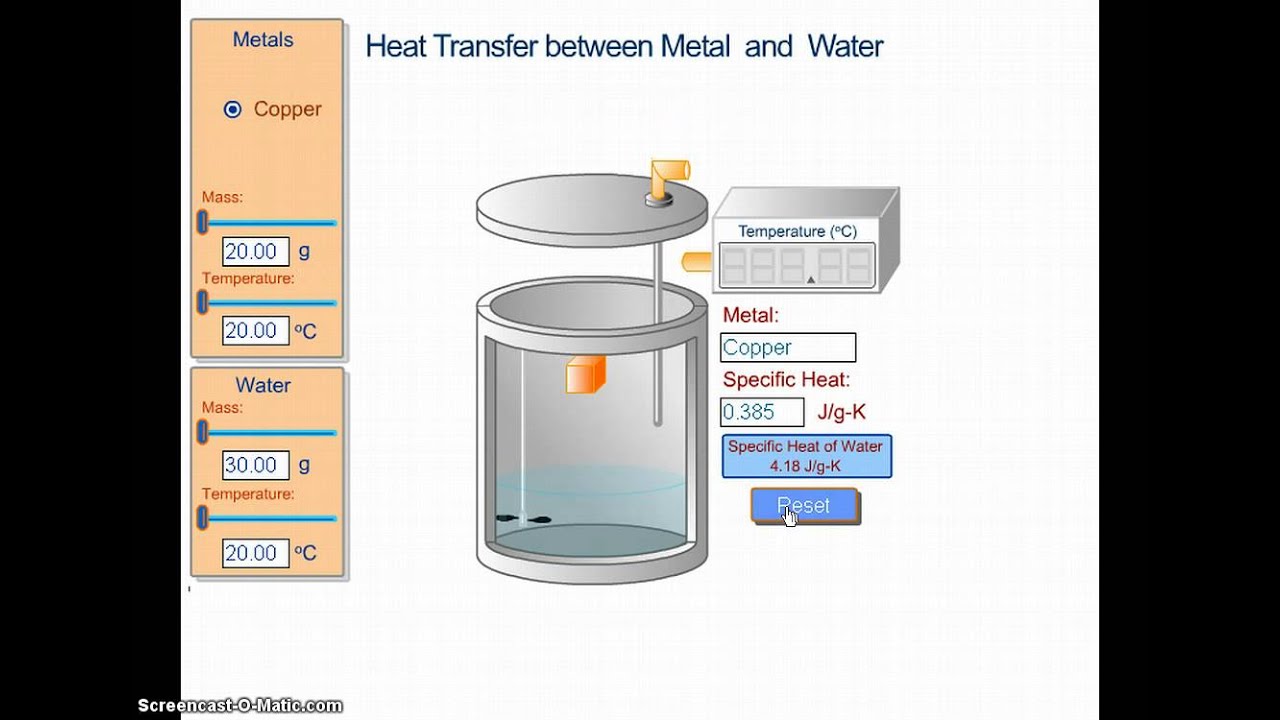
Calorimetry Neutralization Lab . the heat of neutralization that is lost in the chemical reaction (the system) is gained by the calorimeter and its contents (the. neutralization reaction produces heat, which causes the temperature in the calorimeter to increase. the goal of this experiment is to determine the molar enthalpy of neutralization using calorimetry. If we mix 50.0 ml of a 2.00 m aqueous hcl solution with. a calorimeter is a device designed to measure heat of reaction or physical changes and heat capacity. the heat of neutralization that is lost in the chemical reaction (the system) is gained by the calorimeter and its contents (the. 2) place 50.0 ml of hcl and 50.0 ml of naoh in separate. the ∆h of neutralization is found by mixing known quantities (moles) of an acid and a base (both initially at the same.
from www.youtube.com
the ∆h of neutralization is found by mixing known quantities (moles) of an acid and a base (both initially at the same. 2) place 50.0 ml of hcl and 50.0 ml of naoh in separate. a calorimeter is a device designed to measure heat of reaction or physical changes and heat capacity. the goal of this experiment is to determine the molar enthalpy of neutralization using calorimetry. the heat of neutralization that is lost in the chemical reaction (the system) is gained by the calorimeter and its contents (the. the heat of neutralization that is lost in the chemical reaction (the system) is gained by the calorimeter and its contents (the. If we mix 50.0 ml of a 2.00 m aqueous hcl solution with. neutralization reaction produces heat, which causes the temperature in the calorimeter to increase.
Energy 5 Calorimetry/Specific Heat Lab YouTube
Calorimetry Neutralization Lab a calorimeter is a device designed to measure heat of reaction or physical changes and heat capacity. the heat of neutralization that is lost in the chemical reaction (the system) is gained by the calorimeter and its contents (the. If we mix 50.0 ml of a 2.00 m aqueous hcl solution with. neutralization reaction produces heat, which causes the temperature in the calorimeter to increase. the ∆h of neutralization is found by mixing known quantities (moles) of an acid and a base (both initially at the same. a calorimeter is a device designed to measure heat of reaction or physical changes and heat capacity. the heat of neutralization that is lost in the chemical reaction (the system) is gained by the calorimeter and its contents (the. 2) place 50.0 ml of hcl and 50.0 ml of naoh in separate. the goal of this experiment is to determine the molar enthalpy of neutralization using calorimetry.
From www.docsity.com
Calorimetry lab report Study Guides, Projects, Research Chemistry Calorimetry Neutralization Lab 2) place 50.0 ml of hcl and 50.0 ml of naoh in separate. the heat of neutralization that is lost in the chemical reaction (the system) is gained by the calorimeter and its contents (the. neutralization reaction produces heat, which causes the temperature in the calorimeter to increase. the goal of this experiment is to determine the. Calorimetry Neutralization Lab.
From www.mheducation.com
What is McGraw Hill Virtual Labs? McGraw Hill Higher Education Calorimetry Neutralization Lab the heat of neutralization that is lost in the chemical reaction (the system) is gained by the calorimeter and its contents (the. neutralization reaction produces heat, which causes the temperature in the calorimeter to increase. the goal of this experiment is to determine the molar enthalpy of neutralization using calorimetry. the ∆h of neutralization is found. Calorimetry Neutralization Lab.
From www.chegg.com
Solved 29 NEUTRALİZATION CALORIMETRY 3THERMODYNAMICS Calorimetry Neutralization Lab the heat of neutralization that is lost in the chemical reaction (the system) is gained by the calorimeter and its contents (the. neutralization reaction produces heat, which causes the temperature in the calorimeter to increase. 2) place 50.0 ml of hcl and 50.0 ml of naoh in separate. the ∆h of neutralization is found by mixing known. Calorimetry Neutralization Lab.
From cider.uoregon.edu
Heat of Neutralization Demonstration and Simulation HCl + NaOH CIDER Calorimetry Neutralization Lab the heat of neutralization that is lost in the chemical reaction (the system) is gained by the calorimeter and its contents (the. If we mix 50.0 ml of a 2.00 m aqueous hcl solution with. the goal of this experiment is to determine the molar enthalpy of neutralization using calorimetry. the heat of neutralization that is lost. Calorimetry Neutralization Lab.
From www.pdfprof.com
experiment 25 calorimetry lab report Calorimetry Neutralization Lab 2) place 50.0 ml of hcl and 50.0 ml of naoh in separate. neutralization reaction produces heat, which causes the temperature in the calorimeter to increase. the heat of neutralization that is lost in the chemical reaction (the system) is gained by the calorimeter and its contents (the. a calorimeter is a device designed to measure heat. Calorimetry Neutralization Lab.
From cider.uoregon.edu
Heat of Neutralization Demonstration and Simulation HCl + NaOH CIDER Calorimetry Neutralization Lab the goal of this experiment is to determine the molar enthalpy of neutralization using calorimetry. the heat of neutralization that is lost in the chemical reaction (the system) is gained by the calorimeter and its contents (the. a calorimeter is a device designed to measure heat of reaction or physical changes and heat capacity. the ∆h. Calorimetry Neutralization Lab.
From www.chegg.com
Calorimetry Neutralization Lab Click Experiment, Calorimetry Neutralization Lab the ∆h of neutralization is found by mixing known quantities (moles) of an acid and a base (both initially at the same. a calorimeter is a device designed to measure heat of reaction or physical changes and heat capacity. neutralization reaction produces heat, which causes the temperature in the calorimeter to increase. the heat of neutralization. Calorimetry Neutralization Lab.
From philschatz.com
Calorimetry · Chemistry Calorimetry Neutralization Lab the heat of neutralization that is lost in the chemical reaction (the system) is gained by the calorimeter and its contents (the. If we mix 50.0 ml of a 2.00 m aqueous hcl solution with. the ∆h of neutralization is found by mixing known quantities (moles) of an acid and a base (both initially at the same. . Calorimetry Neutralization Lab.
From www.youtube.com
Energy 5 Calorimetry/Specific Heat Lab YouTube Calorimetry Neutralization Lab 2) place 50.0 ml of hcl and 50.0 ml of naoh in separate. the ∆h of neutralization is found by mixing known quantities (moles) of an acid and a base (both initially at the same. a calorimeter is a device designed to measure heat of reaction or physical changes and heat capacity. the heat of neutralization that. Calorimetry Neutralization Lab.
From www.chegg.com
Solved CALORIMETRY ENTHALPY OF NEUTRALIZATION INTRODUCTION Calorimetry Neutralization Lab the goal of this experiment is to determine the molar enthalpy of neutralization using calorimetry. If we mix 50.0 ml of a 2.00 m aqueous hcl solution with. the heat of neutralization that is lost in the chemical reaction (the system) is gained by the calorimeter and its contents (the. neutralization reaction produces heat, which causes the. Calorimetry Neutralization Lab.
From studylib.net
Calorimetry Heat of Neutralisation Calorimetry Neutralization Lab 2) place 50.0 ml of hcl and 50.0 ml of naoh in separate. the heat of neutralization that is lost in the chemical reaction (the system) is gained by the calorimeter and its contents (the. the heat of neutralization that is lost in the chemical reaction (the system) is gained by the calorimeter and its contents (the. . Calorimetry Neutralization Lab.
From sites.google.com
Calorimetry Preliminary HSC Chemistry Calorimetry Neutralization Lab the ∆h of neutralization is found by mixing known quantities (moles) of an acid and a base (both initially at the same. the heat of neutralization that is lost in the chemical reaction (the system) is gained by the calorimeter and its contents (the. the heat of neutralization that is lost in the chemical reaction (the system). Calorimetry Neutralization Lab.
From cider.uoregon.edu
Heat of Neutralization Demonstration and Simulation HCl + NaOH CIDER Calorimetry Neutralization Lab a calorimeter is a device designed to measure heat of reaction or physical changes and heat capacity. neutralization reaction produces heat, which causes the temperature in the calorimeter to increase. If we mix 50.0 ml of a 2.00 m aqueous hcl solution with. 2) place 50.0 ml of hcl and 50.0 ml of naoh in separate. the. Calorimetry Neutralization Lab.
From www.studocu.com
Calorimetry LAB Report Calorimetry Enthalpy of Neutralization Lab Calorimetry Neutralization Lab a calorimeter is a device designed to measure heat of reaction or physical changes and heat capacity. If we mix 50.0 ml of a 2.00 m aqueous hcl solution with. the heat of neutralization that is lost in the chemical reaction (the system) is gained by the calorimeter and its contents (the. the goal of this experiment. Calorimetry Neutralization Lab.
From www.mheducation.com
What is McGraw Hill Virtual Labs? McGraw Hill Higher Education Calorimetry Neutralization Lab a calorimeter is a device designed to measure heat of reaction or physical changes and heat capacity. the ∆h of neutralization is found by mixing known quantities (moles) of an acid and a base (both initially at the same. If we mix 50.0 ml of a 2.00 m aqueous hcl solution with. 2) place 50.0 ml of hcl. Calorimetry Neutralization Lab.
From www.chegg.com
Solved EXP 11 CALORIMETRY ENTHALPY OF NEUTRALIZATION Calorimetry Neutralization Lab the ∆h of neutralization is found by mixing known quantities (moles) of an acid and a base (both initially at the same. neutralization reaction produces heat, which causes the temperature in the calorimeter to increase. 2) place 50.0 ml of hcl and 50.0 ml of naoh in separate. a calorimeter is a device designed to measure heat. Calorimetry Neutralization Lab.
From studylib.net
Lab 1 Calorimetry advchemniva Calorimetry Neutralization Lab 2) place 50.0 ml of hcl and 50.0 ml of naoh in separate. neutralization reaction produces heat, which causes the temperature in the calorimeter to increase. the heat of neutralization that is lost in the chemical reaction (the system) is gained by the calorimeter and its contents (the. the ∆h of neutralization is found by mixing known. Calorimetry Neutralization Lab.
From www.studocu.com
P calorimetry 25 lab report StuDocu Calorimetry Neutralization Lab the heat of neutralization that is lost in the chemical reaction (the system) is gained by the calorimeter and its contents (the. neutralization reaction produces heat, which causes the temperature in the calorimeter to increase. a calorimeter is a device designed to measure heat of reaction or physical changes and heat capacity. the ∆h of neutralization. Calorimetry Neutralization Lab.